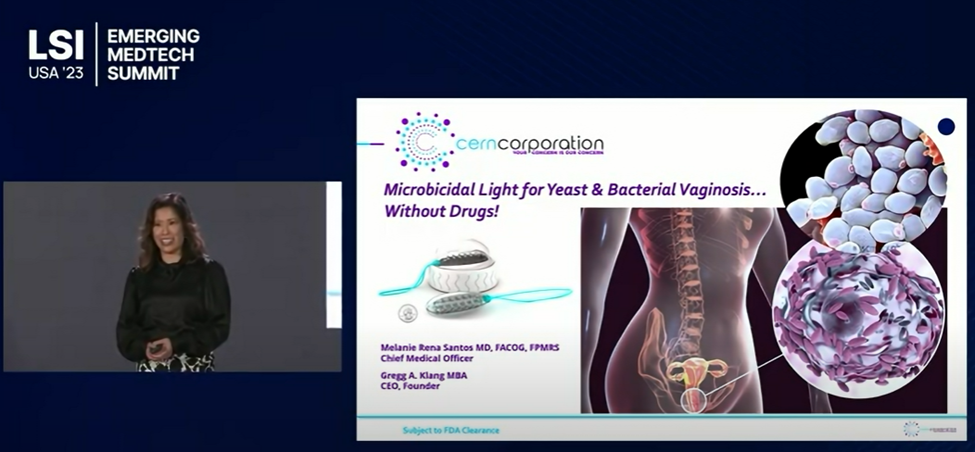
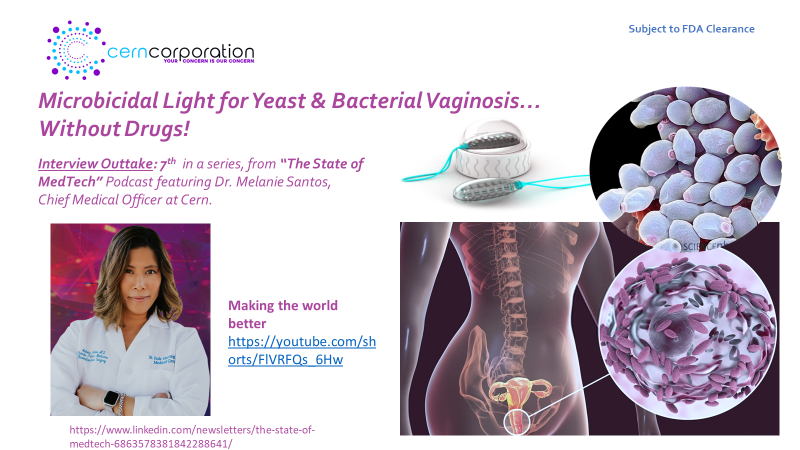
Cern Corporation – Developing a much-needed, non-drug therapy, for mitigation of yeast & bacterial vaginosis (without the need for drugs), is excited to welcome new key members to the team.
Dr. Neal Lonky, OBGYN, MPH – Dr. Lonky is a recognized leader in women’s healthcare occupying significant roles at both Kaiser Permanente as well as The University of California Irvine as a practicing physician, research lead and key investigator. Dr. Lonky has provided significant support to SCMPMG to improve patient care, safety, and affordability and is an acknowledged expert in women’s health policy and management. Additionally, Dr. Lonky is on the Editorial Board of American Society of Colposcopy Journal of Lower Genital Tract Disease and Contributing Editor for OBG Management. Editorial Board of American Society of Colposcopy Journal of Lower Genital Tract Disease and Contributing Editor for OBG Management. Dr. Lonky’s association with Kaiser includes, Practicing Physician, Elected Board of Directors, Chairman of the Quality Committee of the Board of Directors, Oversight of operations and policy for the Southern California Permanente Medical Group in concert with Kaiser Foundation. Co-Chairman of the Elected Board of Directors, Chairman of the Quality Committee of the Board of Directors. Association with The University of California Irvine includes roles as Clinical Professor as well as Faculty Clinical Professor at the Dept. of Obstetrics and Gynecology. Dr. Lonky is also CEO, Founder (and inventor) at Histologics LLC, who’s focus is minimally Invasive tissue sampling and wound hygiene/debridement devices.
“The ability to align this type of significant talent to Cern’s mission, the mitigation of yeast & bacterial vaginosis – without the need for systemic, antifungal/antibiotics, is affirmation as what we are doing for women’s health clearly resonates with subject matter experts in various capacities”. Gregg A. Klang, Cern Corp. CEO and Founder
The Cern Device…For those for whom drug based therapies are neither effective, appropriate, or desired!